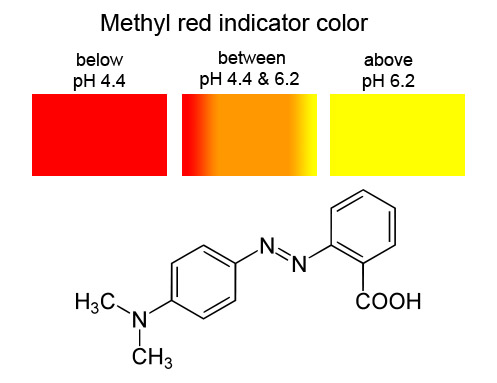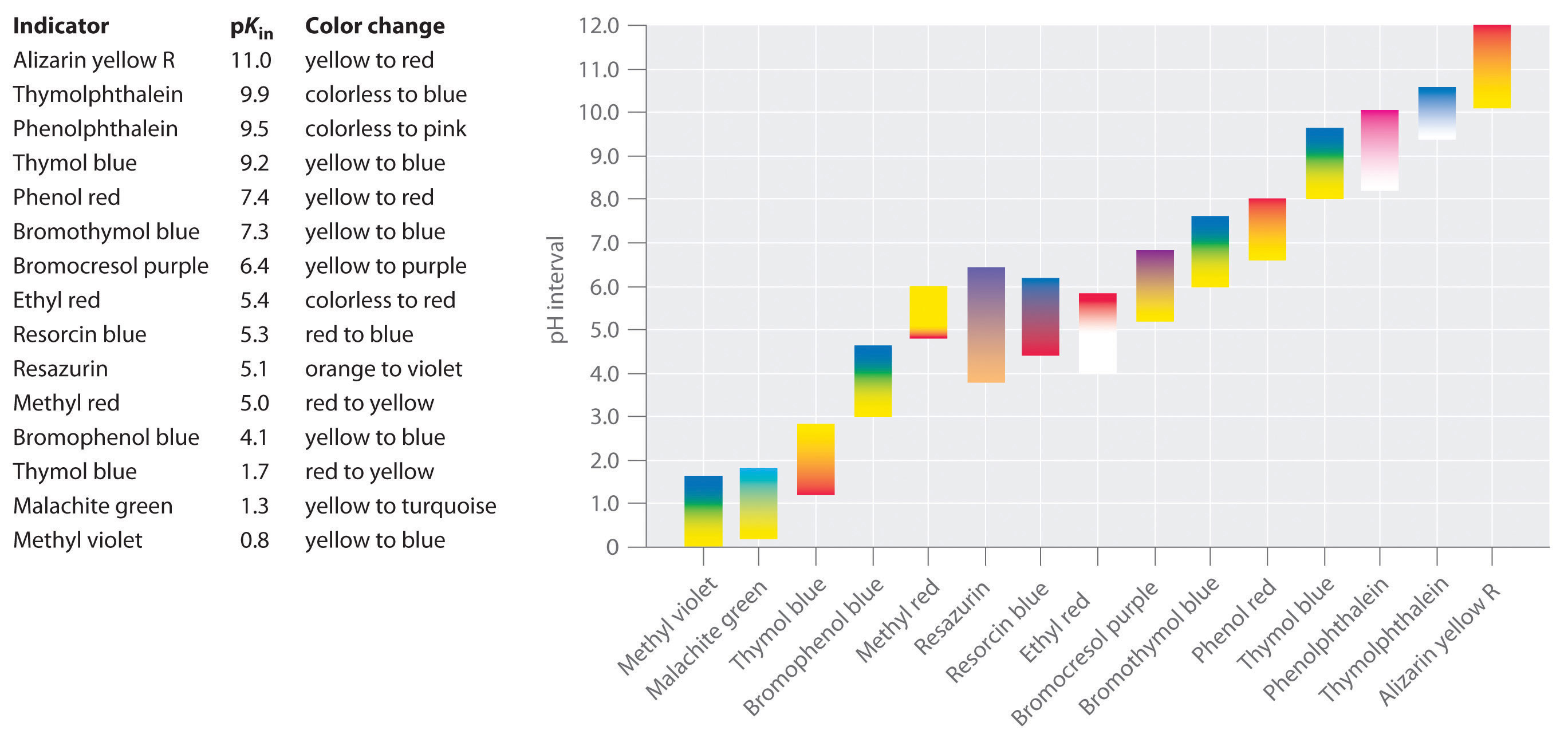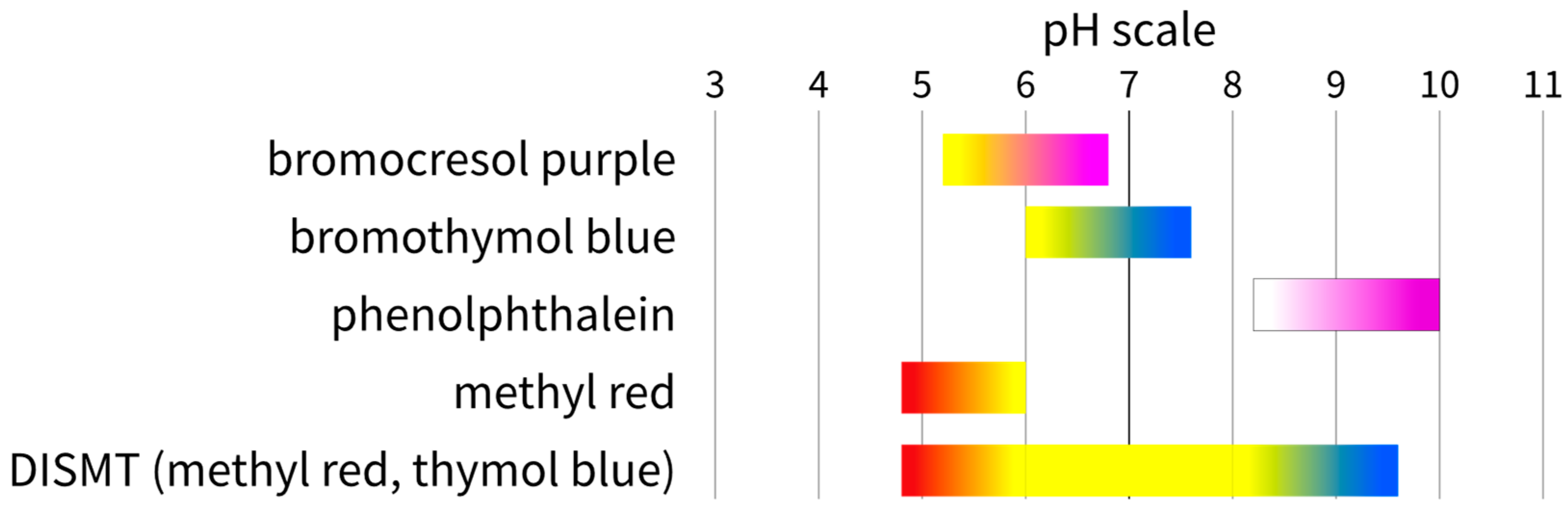
Energies | Free Full-Text | The Colourimetric Method for Mixing Time Measurement in Single-Use and Multi-Use Bioreactors—Methodology Overview and Practical Recommendations

Photograph of color changes of methyl red indicator solutions after... | Download Scientific Diagram

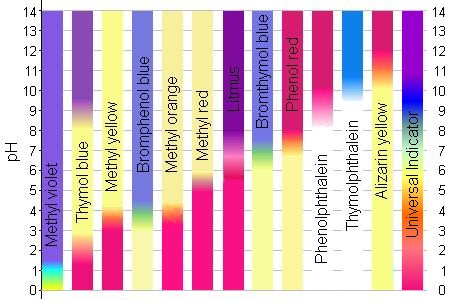
4.1 – Describe how acids and bases in solution affect certain indicators 4.2 Determine the pH of a solution using a universal indicator PH SCALE. - ppt download
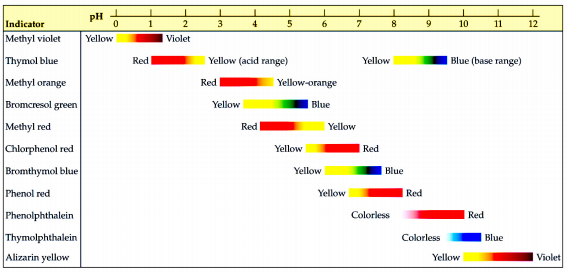
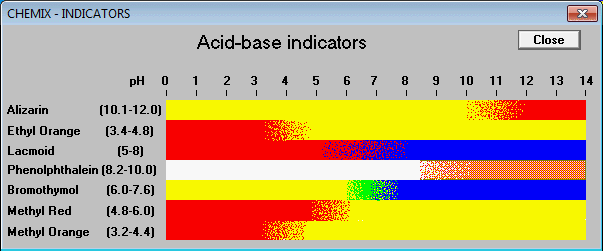
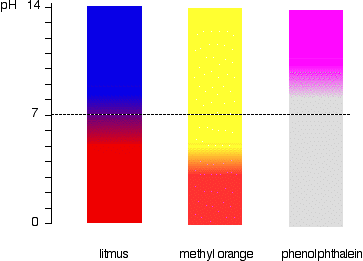
A particular solution produced a yellow colour when tested with both cresol red, methyl orange and bromothymol blue. What does this imply about the pH of the solution? - Quora