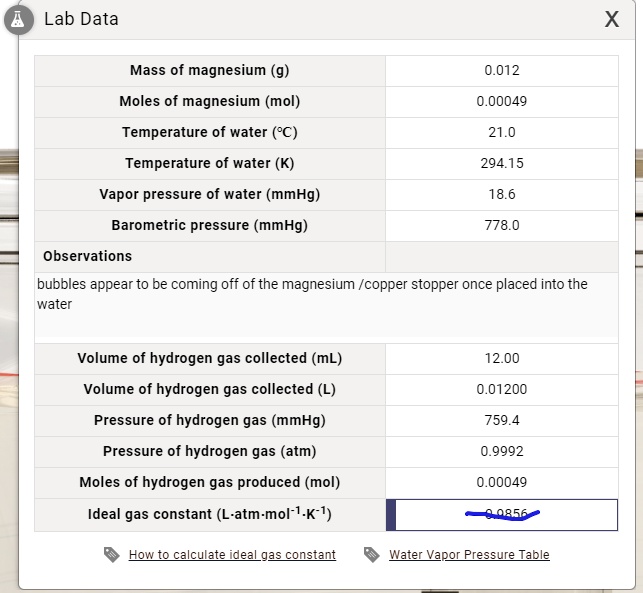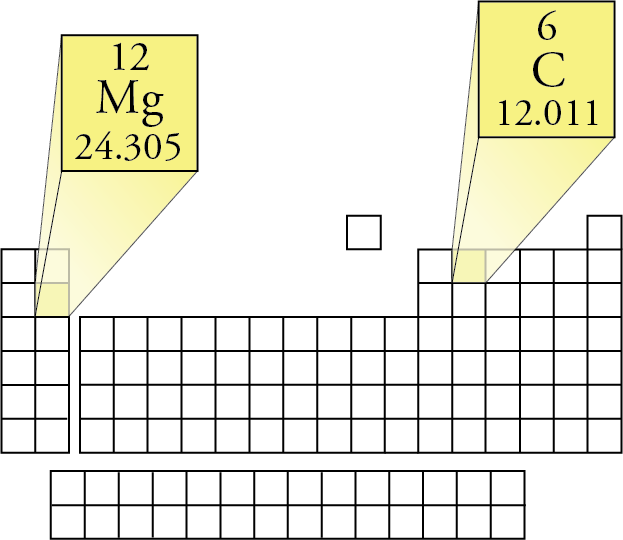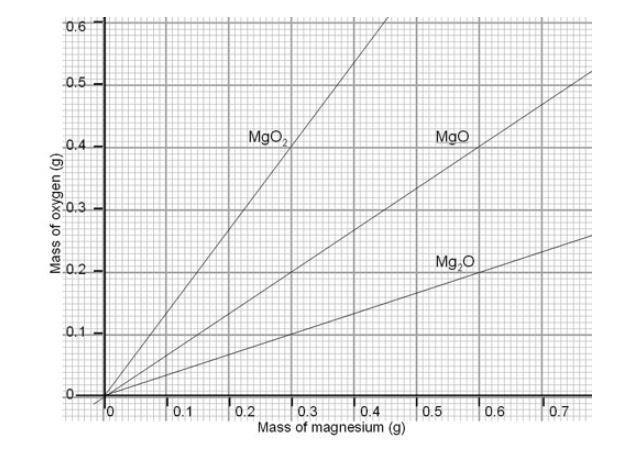
Determination of the molar mass of Magnesium from the reaction of magnesium and hydrochloric acid. - International Baccalaureate Chemistry - Marked by Teachers.com

1a.calculate the relative formula mass of magnesium oxide when the relative atomic masses are O=16 - Brainly.com

In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

The average atomic mass of a mixture containing 79 mole percent of ^24 Mg and remaining 21 mole percent of ^25 Mg and ^26 Mg , is 24.31. Mole percent of ^26 Mg is :

2. Using the following data, calculate the average atomic mass of magnesium (give your answer to the - Brainly.com

SOLVED: Calculatious Mass of magnesium (3-1) 36.79g-36.58g-0.21 36.44g 36.58g-0.36 0.36g-0.21g-0.15 G 0 0.21g/24.3g/mol-0.0086-mole Mass of magnesium compound (4-1) Mass of oxygcn in the product (4-3) Moles ofMg (Show calculations ) Moles of

⚗️calculate the mass of magnesium needed to make 25g of magnesium oxidemark brainliest of correct xx - Brainly.com
Mg Magnesium Chemical Element Periodic Table. Single vector illustration, colorful Icon with molar mass, electron conf. and atomic number Stock Vector Image & Art - Alamy