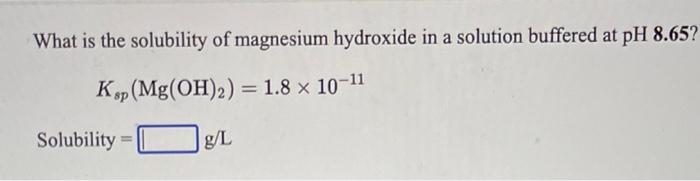If the solubility of Mg(OH)2 in water is 1.4 × 10^-x . Ksp = 1.2 × 10^-11 . Find out the value of x.

Inhibition of Homogeneous Formation of Magnesium Hydroxide by Low-Molar-Mass Poly(acrylic acid) with Different End-Groups | Industrial & Engineering Chemistry Research

Crystallization of calcium carbonate and magnesium hydroxide in the heat exchangers of once-through Multistage Flash (MSF-OT) desalination process - ScienceDirect

SOLVED: Question 66 0 / 1 pts What is the solubility of magnesium hydroxide; Mg(OH)z, if the solubility product is 2.00 x 10-11 (mol/L)3? t Answer 1.71X 10-4 mol/L 3.16 X 10-6
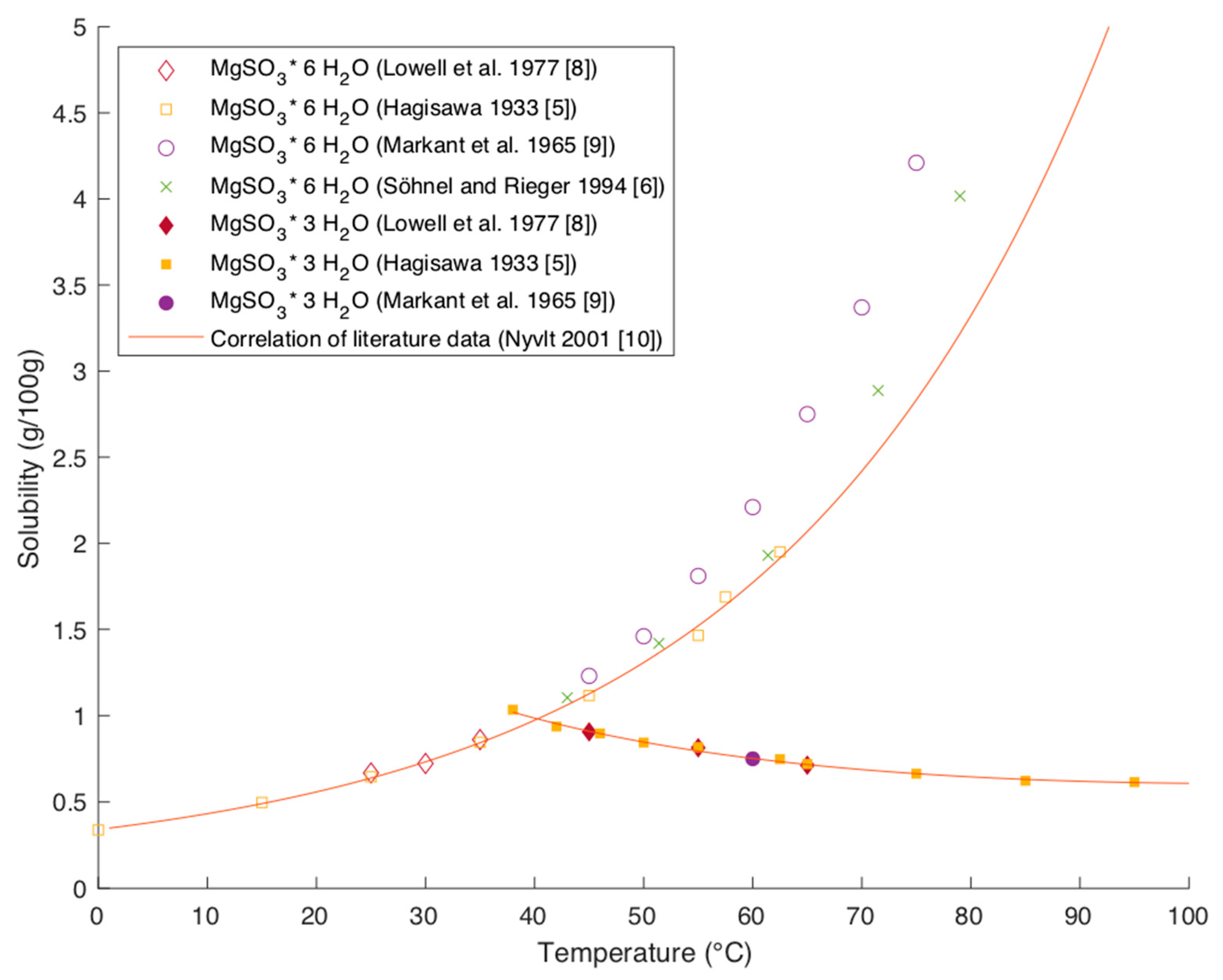
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling

pH of a saturated solution fo magnesium hydroxide in water at `298K` is 10. The solubility - YouTube
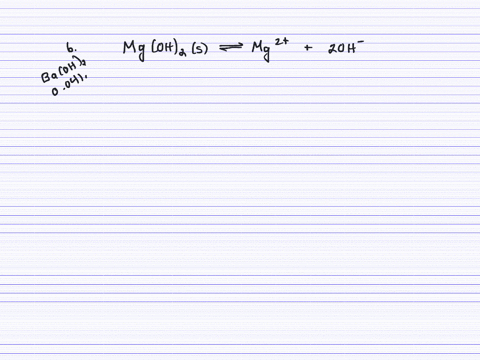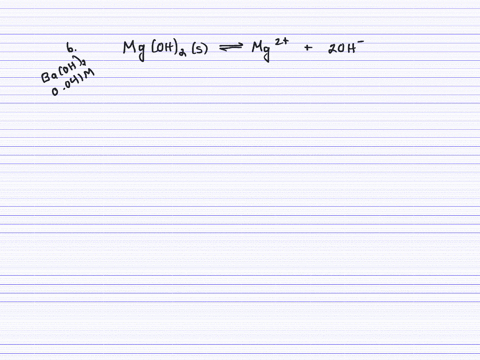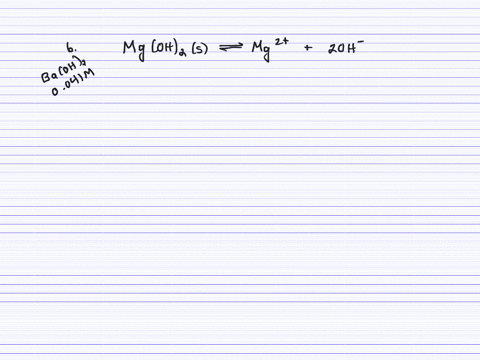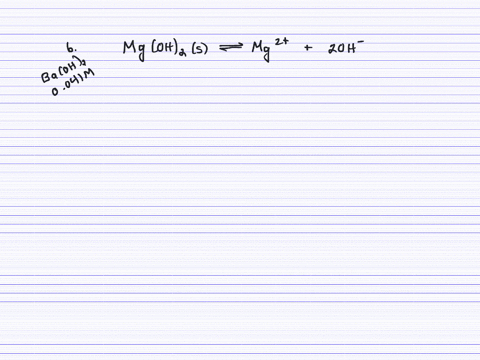
SOLVED:Calculate the solubility (in grams per liter) of magnesium hydroxide in the following. (a) pure water (b) 0.041 M Ba(OH)2 (c) 0.0050 M MgCl2
![SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ] SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ]](https://cdn.numerade.com/ask_images/e1aaca9121da4955a58de8b427babda4.jpg)
SOLVED: Calculate the solubility constant for dissolving Magnesium hydroxide Mg(OH) in water if [Mgl is 0.014 Problem 3: Calculate the solubility constant for dissolving Calcium hydroxide Ca(OH)z in water if [Ca ]