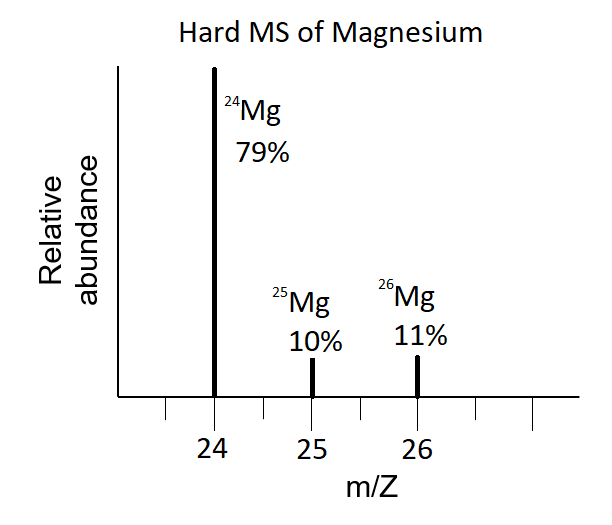
⚗️Magnesium occurs naturally in only three isotopes. ^24 Mg has an isotopic mass of 23.9850 amu and - Brainly.com

SOLVED: There are three naturally-occurring isotopes of magnesium (Mg) The chart below summarizes the pertinent information about these three isotopes Show the calculation for - the average atomic mass of magnesium. Symbol

SOLVED: Naturally occurring magnesium has an atomic mass of 24.312 and consists of three isotopes. The major isotope is 24Mg, natural abundance 78.99%, relative atomic mass 23.98504. The next most abundant isotope

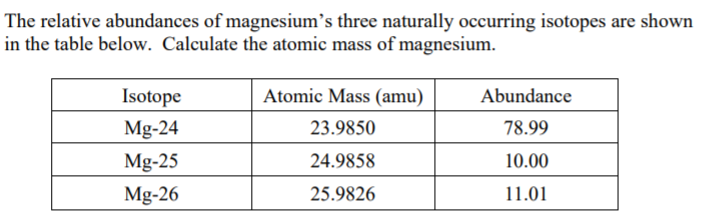
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: \begin{array}{|c|c|c|} \hline \text{Isotope} & \text{Mass (amu)} & \text{Abundance (%)} \\ \hline \text{Mg-24} & \text{23.9850} & \text{78.99} \\ \hline \t

SOLVED: Calculate the average atomic mass of magnesium using the following data for threc magnesium Isotopes Ksotope massl relativg abuudance Mg-24 23.985 0.7870 Mg-25 24.986 0.1013 Mg-26 25,983 0.1417 Calculatc the average

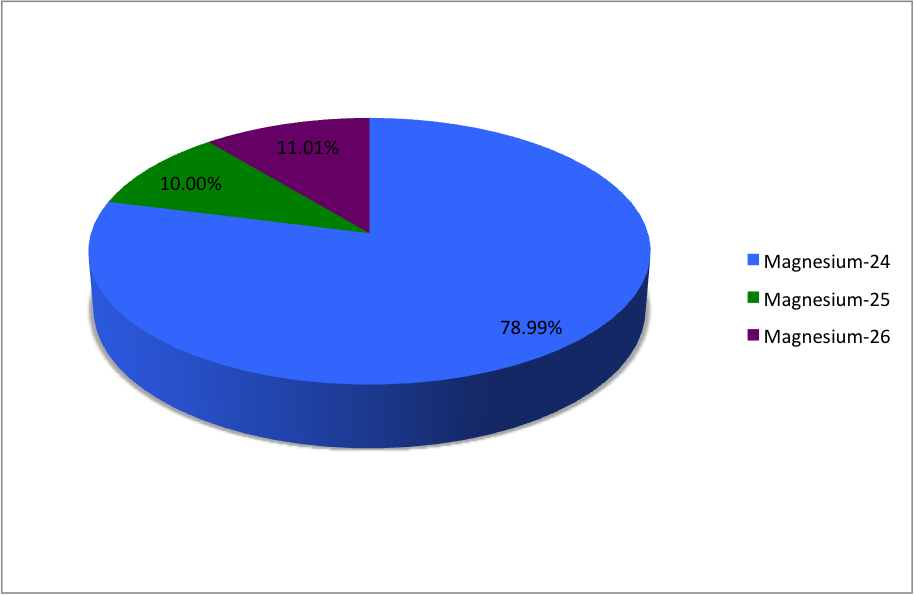
In a periodic table the average atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance on earth. The three isotopes and their

1 Warm Up Isotopes Mass of Isotope Abundance 24 Mg =24.0 amu 78.70% 25 Mg = 25.0 amu 10.13% 26 Mg = 26.0 amu 11.17% Calculate the mass average of magnesium. - ppt download

Surface atomic magnesium abundance of magnesium strips after soaking in... | Download Scientific Diagram
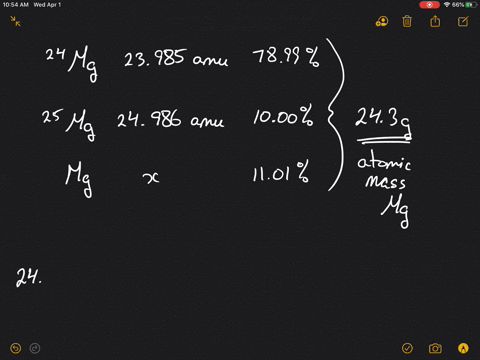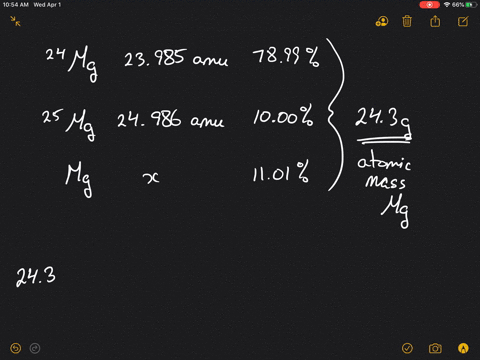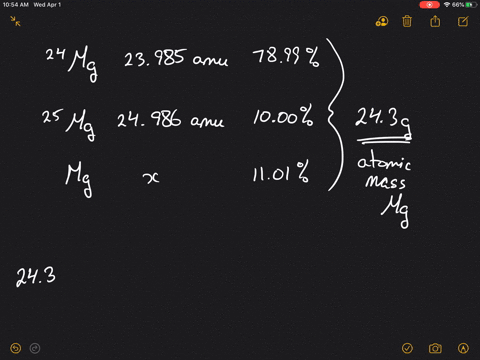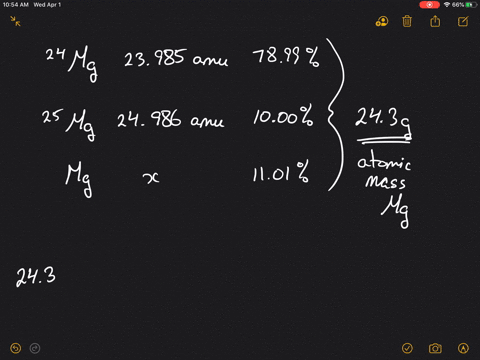
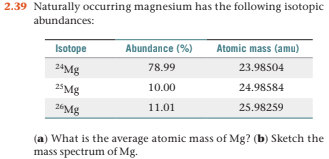
SOLVED:Magnesium has three naturally occurring isotopes: ^24 Mg (23.985 amu) with 78.99% abundance, ^{25} \mathrm{Mg} (24.986 amu) with 10.00% abundance, and a third with 11.01% abundance. Look up the atomic mass of
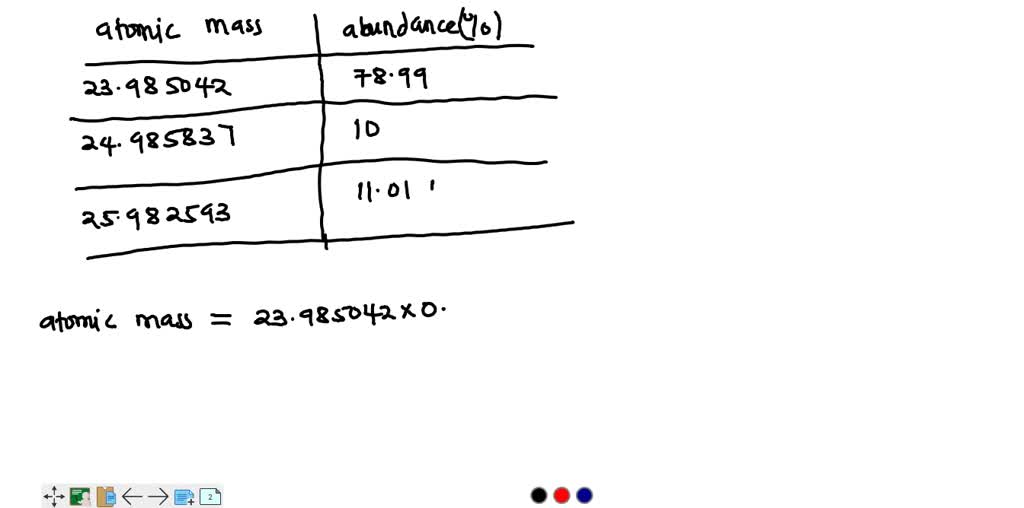
SOLVED: Magnesium has three natural isotopes. Their abundance percentages and masses in nature is as follows: 78.99%; 23.985042 akb, 10.00%; 24.985837 akb, 11.01%; 25.982593 akb. Accordingly, the weighted average atomic mass of magnesium calculate.

Lick index diagram to diagnose the magnesium-to-iron abundance ratio in... | Download Scientific Diagram