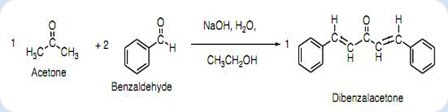
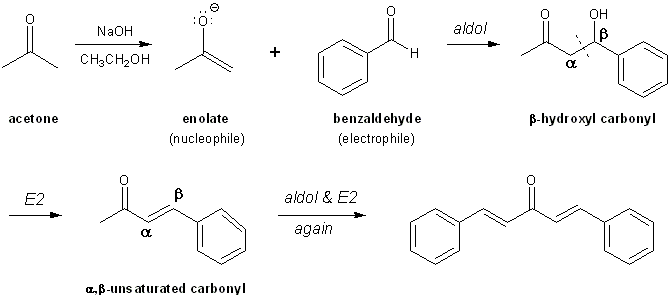
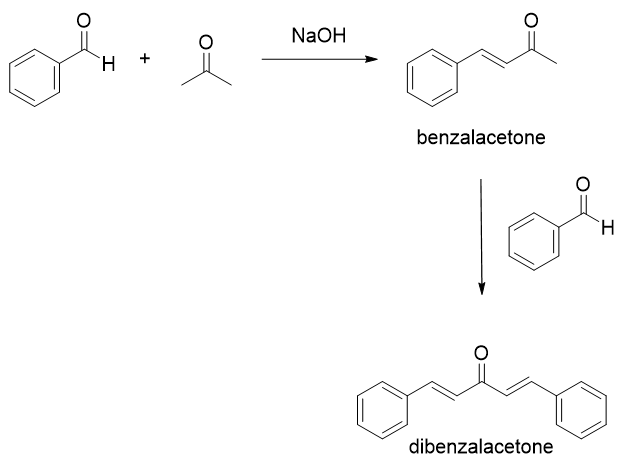
Write a reaction that would take place during the synthesis of dibenzalacetone if the concentration of acetone in the reaction mixture is not carefully controlled. | Homework.Study.com
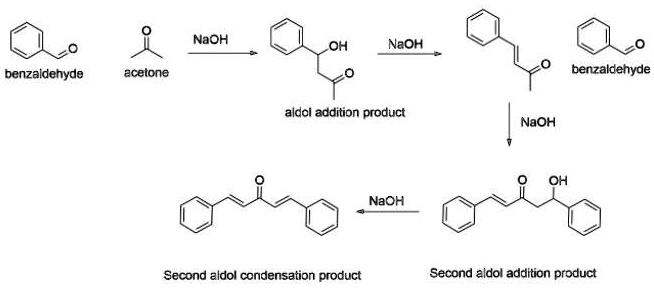
SOLVED: NaOH OH NaOH benzaldehyde acetone benzaldehyde aldal additipn product NaOH OH NaoH Second aldol condensation product Second aldol addition product

Write a reaction that would take place during the synthesis of dibenzalacetone if the concentration of acetone in the reaction mixture is not carefully controlled. | Homework.Study.com

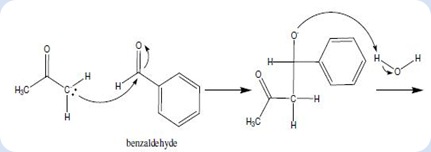
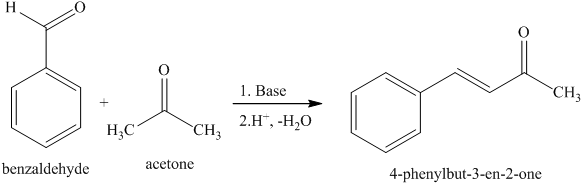
The product formed by aldol condensation between benzaldehyde and acetone is benzalacetone. Its structure is:
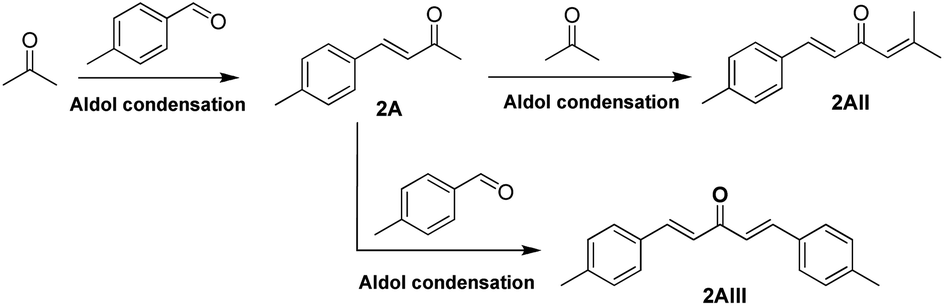
Synthesis of jet fuel range high-density dicycloalkanes with methyl benzaldehyde and acetone - Sustainable Energy & Fuels (RSC Publishing) DOI:10.1039/D0SE01110J
Scheme 1 Mannich reaction between benzaldehyde 1, aniline 2 and acetone 3. | Download Scientific Diagram

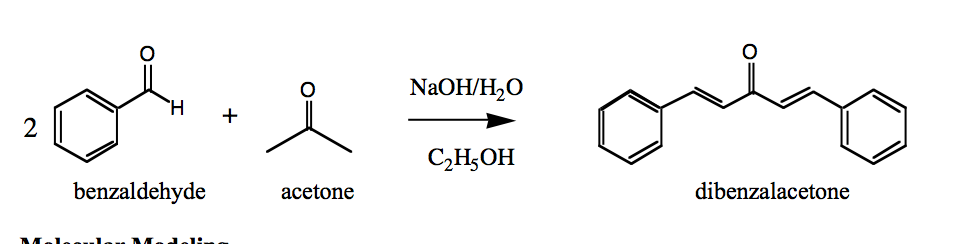
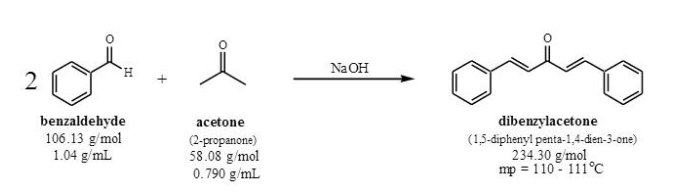
The condensation reaction between one equivalent of acetone and two equivalents of benzaldehyde in presence of dilute alkali leads to the formation of:

Highly tunable selectivity to benzaldehyde over Pd/ZrO2 catalysts in Oppenauer oxidation of benzyl alcohol using acetone as H-acceptor - ScienceDirect